Organization
Division of Molecular Genetics
Staff

Professor
HIRAO, Atsushi

Assistant Professor
TADOKORO, Yuko

Assistant Professor
KOBAYASHI, Masahiko

Assistant Professor
UENO, Masaya

Assistant Professor
Mahmoud Ibrahim Shoulkamy Ibrahim
Aims, Ongoing Projects, and Recent Achievements
Stem cells are defined as cells that have the ability to perpetuate undifferentiated status through self-renewal, and develop into mature cells through differentiation. It has been demonstrated that fine-tuning of self-renewal and differentiation programs, mediated by cooperative networks with intrinsic and extrinsic factors, contributes to stem cell homeostasis in vivo. We have revealed that genes that are involved in longevity, including FOXO and mTOR pathways, contribute to the maintenance of stem cell self-renewal capacity. Thus, signaling pathways for control of intracellular metabolism may play a critical role in stem cell regulation. Furthermore, we have recently identified a molecule that protects hematopoietic homeostasis under diet-induced stress. These findings demonstrate that nutritionassociated signals are critical for determination of stem cell fate.
Dysregulation of self-renewal activity, due to genetic and epigenetic abnormalities, causes tumorigenesis. Acquisition of stem cell property, stemness, promotes malignant progression in cancer. The investigation of distinct and parallel roles in normal stem cells and cancer stem cells will contribute to the design of cancer therapy without damaging normal tissues.
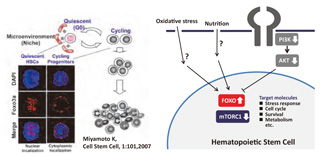 Fig.1 mTOR and FOXO pathways in quiescent hematopoietic stem cells
Fig.1 mTOR and FOXO pathways in quiescent hematopoietic stem cells
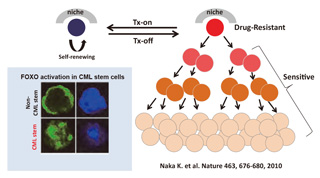 Fig.2 FOXO activation for drug-resistance of leukemia stem cells
Fig.2 FOXO activation for drug-resistance of leukemia stem cells
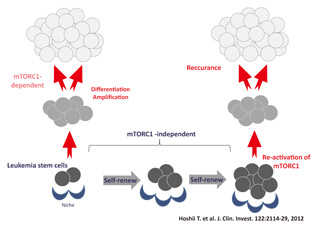 Fig.3 mTOR complex in leukemia stem cells
Fig.3 mTOR complex in leukemia stem cells
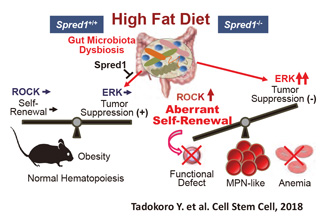 Fig.4 Spred1 as a safeguard of hematopoietic homeostasis against high-fat diet
Fig.4 Spred1 as a safeguard of hematopoietic homeostasis against high-fat diet

